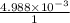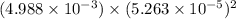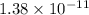Chemistry, 28.01.2020 01:31 SchoolSucks234
The solubility product of calcium fluoride (caf2(s); fluorite) is 310-11 at 25c. could a fluoride concentration of 1.0 mg l-1 be obtained in water that contains 200 mg l-1 of calcium?

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 00:10
In as 1°, 2°, 3°, or 4°. be to . : °b: °c: °d: ° : °b: °c: °d: ° : °b: °c: °d: °e: °f: °g: °h: ° : °b: °c: °d: °e: °f: °g: °h: °i: °
Answers: 3
You know the right answer?
The solubility product of calcium fluoride (caf2(s); fluorite) is 310-11 at 25c. could a fluoride...
Questions






Computers and Technology, 20.12.2019 05:31





Computers and Technology, 20.12.2019 05:31









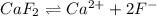
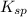 will be as follows.
will be as follows.![K_{sp} = [Ca^{2+}][F^{-}]^{2}](/tpl/images/0474/4010/1d9c8.png)
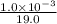
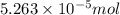
![[F^{-}] = \frac{\text{moles of F^{-}}{volume}](/tpl/images/0474/4010/e0719.png)
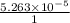
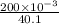
![[Ca^{2+}] = \frac{moles of Ca^{2+}}{volume}](/tpl/images/0474/4010/ce6ee.png)