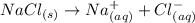When an ionic compound dissolves in water:
a. the negative ends of water molecules surround the positive ions.
b. the negative ends of water molecules surround both the negative and the positive ions.
c. the positive ends of water molecules surround the positive ions.
d. the negative ends of water molecules surround the negative ions.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
When an ionic compound dissolves in water:
a. the negative ends of water molecules surround...
a. the negative ends of water molecules surround...
Questions


Mathematics, 03.08.2019 14:30


Advanced Placement (AP), 03.08.2019 14:30



Computers and Technology, 03.08.2019 14:30

Biology, 03.08.2019 14:30

English, 03.08.2019 14:30

English, 03.08.2019 14:30

History, 03.08.2019 14:30




Mathematics, 03.08.2019 14:30



History, 03.08.2019 14:30