
Chemistry, 29.01.2020 02:51 jamiecoolgal8697
Caffeine (c_8h_10n_4o_2) is a weak base with a k_b value of 4 times 10^-4. the ph of a 0.01 m solution of caffeine is in the range of: a. 2-3 b. 5-6 c. 7-8 d. 9-10 e. 11-12

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Monkeys and bats have similar bone structure in their forelimbs. however, monkeys have longer forelimbs to use for climbing and swinging in trees. bats have shorter forelimbs to use for flight. which term best describes how monkey and bat forelimbs are related to each other? a. homologous b. embryonic c. analogous d. vestigial
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2
You know the right answer?
Caffeine (c_8h_10n_4o_2) is a weak base with a k_b value of 4 times 10^-4. the ph of a 0.01 m soluti...
Questions

Mathematics, 23.10.2019 02:30

Biology, 23.10.2019 02:30

History, 23.10.2019 02:30


Mathematics, 23.10.2019 02:30


History, 23.10.2019 02:30

Mathematics, 23.10.2019 02:30



Mathematics, 23.10.2019 02:30

Mathematics, 23.10.2019 02:30

Mathematics, 23.10.2019 02:30

Mathematics, 23.10.2019 02:30

Mathematics, 23.10.2019 02:30





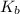 for caffine =
for caffine = 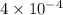
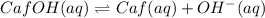
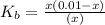
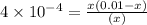
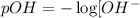
![pOH=-\log[x]](/tpl/images/0479/8766/fba19.png)
![pOH=-\log[0.0096]](/tpl/images/0479/8766/44a89.png)


