Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3


Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1
You know the right answer?
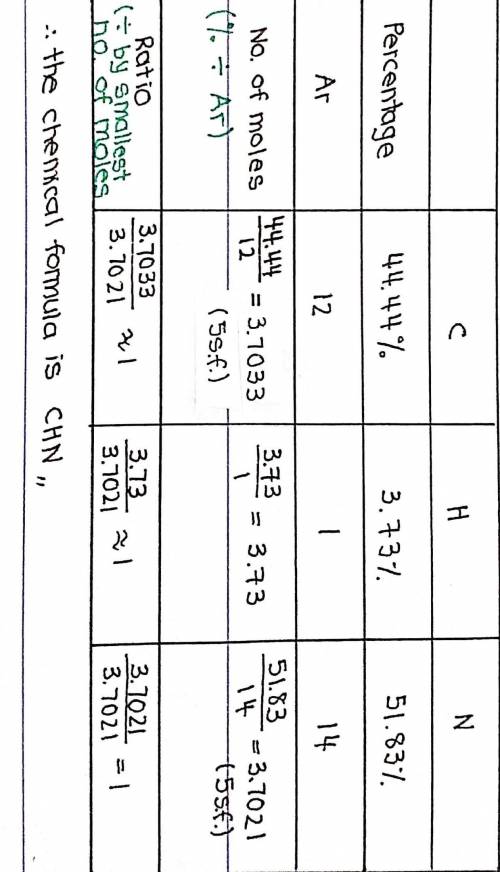
Chemical formula of C 44.44% H 3.73% N 51.83%...
Questions


Mathematics, 23.08.2019 00:30




Computers and Technology, 23.08.2019 00:30

Arts, 23.08.2019 00:30

Biology, 23.08.2019 00:30

Mathematics, 23.08.2019 00:30



Biology, 23.08.2019 00:30



History, 23.08.2019 00:30



Mathematics, 23.08.2019 00:30


Geography, 23.08.2019 00:30