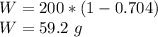Chemistry, 11.02.2020 03:37 giordanolucia18
In 200 g of a concentrated solution of 70.4 wt% nitric acid (r = 1.41 g/mL, FW(HNO3) = 63.01 g/mol), how many grams of water are there in this solution? Assume that the only components of the mixture are water and nitric acid.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
There is a single path for electrons. the current decreases when additional resistors are added. the current will be the same in each resistor. these statements best describe a(n) circuit.
Answers: 3



Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1
You know the right answer?
In 200 g of a concentrated solution of 70.4 wt% nitric acid (r = 1.41 g/mL, FW(HNO3) = 63.01 g/mol),...
Questions


Mathematics, 05.09.2020 22:01

Mathematics, 05.09.2020 22:01


Arts, 05.09.2020 22:01










Chemistry, 05.09.2020 22:01


Chemistry, 05.09.2020 22:01


Social Studies, 05.09.2020 22:01