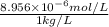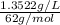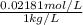Chemistry, 12.02.2020 02:17 ethangorrell67
High concentrations of ammonia (NH3), nitrite ion, and nitrate ion in water can kill fish. Lethal concentrations of these species for rainbow trout are approximately 1.002 mg/L, 0.412 mg/L, and 1352.2 mg/L, respectively. Express these concentrations in molality units, assuming a solution density of 1.00 g/mL. a. m ammoniab. m nitrite ironc. m nitrate ion

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
If 200.0g of copper(ll) sulfate react with an excess of zinc metal, what is the theoretical yield of copper
Answers: 1

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2

You know the right answer?
High concentrations of ammonia (NH3), nitrite ion, and nitrate ion in water can kill fish. Lethal co...
Questions



Mathematics, 24.09.2020 14:01


Mathematics, 24.09.2020 14:01






Mathematics, 24.09.2020 14:01

Engineering, 24.09.2020 14:01

Advanced Placement (AP), 24.09.2020 14:01

Computers and Technology, 24.09.2020 14:01






Spanish, 24.09.2020 14:01

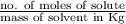
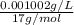
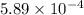 mol/L
mol/L
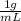 = 1 kg/L
= 1 kg/L
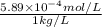
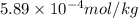
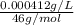
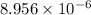 mol/L
mol/L