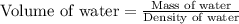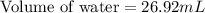Chemistry, 12.02.2020 04:41 carlosgc19
A student obtained a clean, dry, glass-stoppered flask. He weighed the flask stopper and found the total mass to be 32.634g. He then filled the flask with water, weighed again, and obtained a mass of 59.479g. At the temperature of the water, he found that its density was 0.9973 g/mL. a.) What was the mass of the water? (show work)b.) What was the volume of the water? (Show work)c.) What was the volume of the flask? (show work)

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 03:00
Asample of sea water contains 6.28g of sodium chloride per litre of solution. how many milligrams of sodium chloride would be contained in 15.0ml of this solution?
Answers: 3

Chemistry, 23.06.2019 11:30
Which of the following is a property of nonmetals? a.nonmetals are ductile. b.nonmetals have a shiny luster. c.nonmetals have high density. d.nonmetals are nonconductors.
Answers: 1

Chemistry, 23.06.2019 12:30
What would happen to a weak base dissociation equilibrium if more products we added
Answers: 1
You know the right answer?
A student obtained a clean, dry, glass-stoppered flask. He weighed the flask stopper and found the t...
Questions




Computers and Technology, 26.11.2019 00:31


Health, 26.11.2019 00:31




Chemistry, 26.11.2019 00:31


Computers and Technology, 26.11.2019 00:31