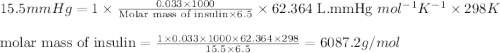Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
Motivation cannot be developed with practice; a person either possesses it or they do not.
Answers: 1

Chemistry, 22.06.2019 00:40
Which is a difference between molecular compounds and ionic compounds? select the correct answer below: question 5 options: molecular compounds typically form between a metal and a nonmetal, while ionic compounds typically form between nonmetals. molecular compounds result from the transfer of electrons between atoms to form ions, while ionic compounds result from the sharing of electrons between neutral atoms. molecular compounds are formed of discrete, neutral molecules, while ionic compounds are formed of large repeating arrays of opposite charges. molecular compounds have high melting points and high boiling points, while ionic
Answers: 3

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2

Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3
You know the right answer?
A solution made by dissolving 33 mg of insulin in 6.5 mL of water has an osmotic pressure of 15.5 mm...
Questions

Mathematics, 26.07.2019 21:20





Mathematics, 26.07.2019 21:20


Mathematics, 26.07.2019 21:20


Mathematics, 26.07.2019 21:20




Mathematics, 26.07.2019 21:20






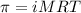
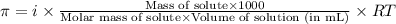
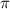 = osmotic pressure of the solution = 15.5 mmHg
= osmotic pressure of the solution = 15.5 mmHg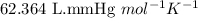
![25^oC=[273+25]=298K](/tpl/images/0508/1937/6a9f9.png)