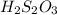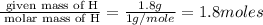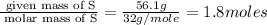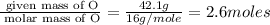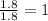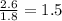A laboratory analysis of a 100 g sample finds it is composed of 1.8 g hydrogen, 56.1 g sulfur, and 42.1 g oxygen. What is its empirical formula? Give your answer in the form H#S#O#, where the number following the element’s symbol corresponds to the subscript in the formula. (Don’t include a 1 subscript explicitly.) For example, the formula CHO would be entered as CH2O.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 19:50
What is the wavelength of a wave with a velocity of 50 m/s and a frequency of 5hz a 250 m b 0.1 m c 10m d 0.01 m
Answers: 2
You know the right answer?
A laboratory analysis of a 100 g sample finds it is composed of 1.8 g hydrogen, 56.1 g sulfur, and 4...
Questions



History, 05.02.2020 03:51

Mathematics, 05.02.2020 03:51



English, 05.02.2020 03:51



Mathematics, 05.02.2020 03:51

Mathematics, 05.02.2020 03:51