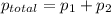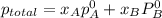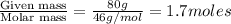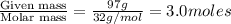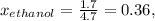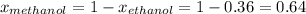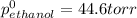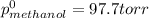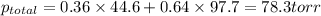Chemistry, 12.02.2020 05:28 usagimiller
At 293 K, methanol has a vapor pressure of 97.7 Torr and ethanol has a vapor pressure of 44.6 Torr. What would be the vapor pressure of a mixture of 80 g of ethanol and 97 g of methanol at 293 K?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:40
In an effort to address concerns about global warming, a power plant in portland,oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1
You know the right answer?
At 293 K, methanol has a vapor pressure of 97.7 Torr and ethanol has a vapor pressure of 44.6 Torr....
Questions

Chemistry, 06.05.2021 20:00

Biology, 06.05.2021 20:00

Mathematics, 06.05.2021 20:00


Biology, 06.05.2021 20:00


English, 06.05.2021 20:00



English, 06.05.2021 20:00





Social Studies, 06.05.2021 20:00


Mathematics, 06.05.2021 20:00



Mathematics, 06.05.2021 20:00

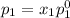 and
and 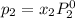
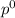 = pressure in the pure state
= pressure in the pure state