Chemistry, 12.02.2020 05:48 cristykianpour
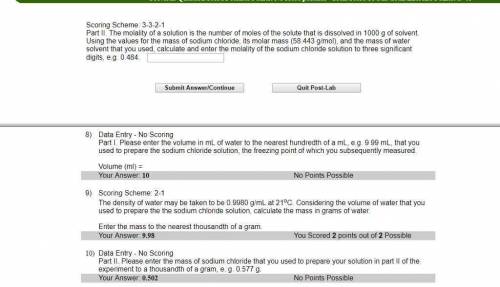
Part II. The molality of a solution is the number of moles of the solute that is dissolved in 1000 g of solvent. Using the values for the mass of sodium chloride, its molar mass (58.443 g/mol), and the mass of water solvent that you used, calculate and enter the molality of the sodium chloride solution to three significant digits, e. g. 0.484.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1
You know the right answer?
Part II. The molality of a solution is the number of moles of the solute that is dissolved in 1000 g...
Questions


Physics, 19.10.2019 16:30

English, 19.10.2019 16:30

English, 19.10.2019 16:30


History, 19.10.2019 16:30



History, 19.10.2019 16:30

Mathematics, 19.10.2019 16:30






Geography, 19.10.2019 16:30

Health, 19.10.2019 16:30



Mathematics, 19.10.2019 16:30