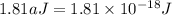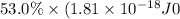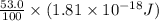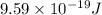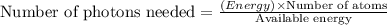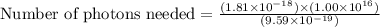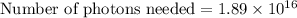Chemistry, 13.02.2020 04:09 blakeley7785
Assuming an ionization efficiency of 53.0 % , how many such photons are needed to ionize 1.00 × 10 16 atoms?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1


Chemistry, 22.06.2019 20:50
What is the vapor pressure of a solution with a benzene to octane?
Answers: 2
You know the right answer?
Assuming an ionization efficiency of 53.0 % , how many such photons are needed to ionize 1.00 × 10 1...
Questions

Social Studies, 28.05.2020 23:05


Mathematics, 28.05.2020 23:05


Biology, 28.05.2020 23:05



Mathematics, 28.05.2020 23:05

Mathematics, 28.05.2020 23:05


Mathematics, 28.05.2020 23:05



Mathematics, 28.05.2020 23:05


English, 28.05.2020 23:05

History, 28.05.2020 23:05



Mathematics, 28.05.2020 23:05