
Chemistry, 13.02.2020 20:02 connorgking
A. Based on the activation energies and frequency factors, rank the following reactions from fastest to slowest reaction rate, assuming they are all at the same temperature and that each starts with the same initial concentration.
E, 50 kJ/mol E,-350 kJ/mol 50 kJ/mol
A = 1.5 × 10-7 s-i A = 1.9 × 10-7 s-i A = 1.5 × 10-7 s-1
Fraction of molecules
The exponential term in the Arrhenius equation is equal to the fraction of molecules, f, with kinetic energy greater than or equal to the activation energy: f=e?Ea/(R?T). Most scientific calculators have an exfunction as the second function of the LN button.
B. A certain reaction with an activation energy of 165 kJ/mol was run at 505 K and again at 525 K . What is the ratio of f at the higher temperature to f at the lower temperature?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1
You know the right answer?
A. Based on the activation energies and frequency factors, rank the following reactions from fastest...
Questions

English, 29.07.2019 22:00




Mathematics, 29.07.2019 22:00

Mathematics, 29.07.2019 22:00

Mathematics, 29.07.2019 22:00






Spanish, 29.07.2019 22:00

Mathematics, 29.07.2019 22:00

Computers and Technology, 29.07.2019 22:00




History, 29.07.2019 22:00

English, 29.07.2019 22:00

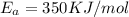 ,
, 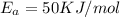 ,
, 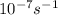 , A = 1.9×
, A = 1.9×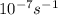 , A=1.5×
, A=1.5×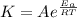
 = Activation Energy
= Activation Energy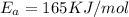
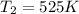
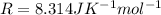
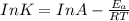
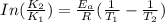
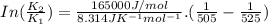
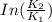 = 19846.04×7.544×
= 19846.04×7.544×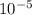 = 1.497
= 1.497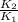 =
=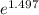 = 4.469
= 4.469 

