Chemistry, 14.02.2020 18:25 Chynadoll94
The product of a Lewis acid-base reaction is a(n) , a single species that contains a new covalent bond. The Lewis acid-base definition focuses on the donation or acceptance of an electron pair to form a new covalent bond in an adduct, the product of an acid-base reaction. Lewis bases the electron pair, and Lewis acids it.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 22.06.2019 17:00
The biosphere of the earth is made up of what compound? organic or inorganic?
Answers: 3

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
The product of a Lewis acid-base reaction is a(n) , a single species that contains a new covalent bo...
Questions

Computers and Technology, 16.07.2020 01:01




English, 16.07.2020 01:01


Mathematics, 16.07.2020 01:01


Chemistry, 16.07.2020 01:01




Biology, 16.07.2020 01:01




Social Studies, 16.07.2020 01:01


World Languages, 16.07.2020 01:01

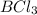 (B has vacant 2p orbital for electron acceptance)
(B has vacant 2p orbital for electron acceptance)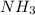 (N has energetically available lone pair)
(N has energetically available lone pair)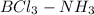 ) where a new covalent bond is formed between B and N.
) where a new covalent bond is formed between B and N.