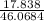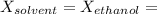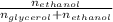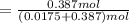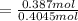Glycerol (C3H8O3), also called glycerine, is widely used in the food and pharmaceutical industries. Glycerol is polar and dissolves readily in water and polar organic solvents like ethanol. Calculate the mole fraction of the solvent in a solution that contains 1.61 g glycerol dissolved in 22.60 mL ethanol (CH3CH2OH; density?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

You know the right answer?
Glycerol (C3H8O3), also called glycerine, is widely used in the food and pharmaceutical industries....
Questions

Mathematics, 20.05.2020 16:58


English, 20.05.2020 16:58

Biology, 20.05.2020 16:58


Mathematics, 20.05.2020 16:58



History, 20.05.2020 16:58

History, 20.05.2020 16:58

Chemistry, 20.05.2020 16:58

Mathematics, 20.05.2020 16:58

Mathematics, 20.05.2020 16:58

Geography, 20.05.2020 16:58

Mathematics, 20.05.2020 16:58



Physics, 20.05.2020 16:58

Physics, 20.05.2020 16:58

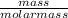
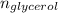 ) =
) = 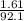
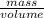
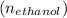 =
=