
Chemistry, 15.02.2020 00:44 juliopejfuhrf5447
The turnover number is defined as the maximum number of substrate molecules that can be converted into product molecules per unit time by an enzyme molecule. The concentration of enzyme active sites is not necessarily equal to the concentration of enzyme molecules, because some enzyme molecules have more than one active site. If the enzyme molecule has one active site, the turnover number is given by turnover number = R max [ E ] t = k 2 ( R max is often written as V max ) If the enzyme molecule has more than one active site, then [ E ] t is multiplied by the number of active sites to determine its effective concentration. Determine the value of the turnover number of the enzyme carbonic anhydrase, given that R max for carbonic anhydrase equals 249 μmol ⋅ L − 1 ⋅ s − 1 and [ E ] t = 2.35 nmol ⋅ L − 1 . Carbonic anhydrase has a single active site.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 21.06.2019 23:00
Plz choose one of the compounds from the table and explain how you know the numbers of atoms in your formula. is it possible for two different compounds to be made from the exact same two elements? why or why not? with a limited number of elements (less than 120 are known), does this mean we also have a small number of compounds or do we have a large number of compounds in this world?
Answers: 1

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1
You know the right answer?
The turnover number is defined as the maximum number of substrate molecules that can be converted in...
Questions

Mathematics, 17.02.2021 19:40

Chemistry, 17.02.2021 19:40

Mathematics, 17.02.2021 19:40




Spanish, 17.02.2021 19:40


English, 17.02.2021 19:40


Computers and Technology, 17.02.2021 19:40




Business, 17.02.2021 19:40

Social Studies, 17.02.2021 19:40


Mathematics, 17.02.2021 19:40

Mathematics, 17.02.2021 19:40

Chemistry, 17.02.2021 19:40

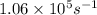
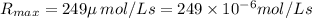
![[E]_t=235 n \, mol\L=235 \times 10^{-9} mol/L](/tpl/images/0512/0801/98a71.png)
![n_{turnover}=\frac{R_{max}}{[E]_t}](/tpl/images/0512/0801/c420f.png)
![n_{turnover}=\frac{R_{max}}{[E]_t}\\n_{turnover}=\frac{249 \times 10^{-6} mol/Ls}{2.35 \times 10^{-9} mol/L}\\n_{turnover}=1.06\times10^5 s^{-1}](/tpl/images/0512/0801/6df3d.png)


