
Chemistry, 15.02.2020 04:27 lekaje2375
A generic element, G, is composed of two isotopes, 132G and 128G. 132G has a natural abundance of 90% and an isotopic mass of 131.90 amu, and 128G has a natural abundance of 10% and an isotopic mass of 127.90 amu. What is the average atomic mass of this element?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2


Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1
You know the right answer?
A generic element, G, is composed of two isotopes, 132G and 128G. 132G has a natural abundance of 90...
Questions






Mathematics, 20.11.2019 02:31

Mathematics, 20.11.2019 02:31


Mathematics, 20.11.2019 02:31


Health, 20.11.2019 02:31


History, 20.11.2019 02:31



History, 20.11.2019 02:31

Mathematics, 20.11.2019 02:31

English, 20.11.2019 02:31


Mathematics, 20.11.2019 02:31

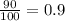
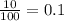
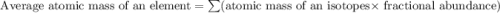
![A=\sum[(131.90\times 0.9)+(127.90\times 0.1)]=131.5amu](/tpl/images/0512/2891/2177c.png)


