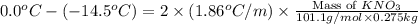Chemistry, 17.02.2020 17:59 Joshuafranklindude
Assuming complete dissociation of the solute, how many grams of KNO3 must be added to 275 mL of water to produce a solution that freezes at −14.5 ∘C? The freezing point for pure water is 0.0 ∘C and Kf is equal to 1.86 ∘C/m .

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 21:00
Two nails have identical sizes and shapes. in one nail, 20 percent of the domains are lined up. in the other nail, 80 percent of the domains are lined up. which has stronger magnetic force? first answer gets brainliest!
Answers: 1

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Assuming complete dissociation of the solute, how many grams of KNO3 must be added to 275 mL of wate...
Questions

Mathematics, 10.03.2021 14:00


Business, 10.03.2021 14:00

Mathematics, 10.03.2021 14:00


World Languages, 10.03.2021 14:00

SAT, 10.03.2021 14:00

Chemistry, 10.03.2021 14:00

Spanish, 10.03.2021 14:00

History, 10.03.2021 14:00


Biology, 10.03.2021 14:00


Mathematics, 10.03.2021 14:00

Mathematics, 10.03.2021 14:00


English, 10.03.2021 14:00


Mathematics, 10.03.2021 14:00

Chemistry, 10.03.2021 14:00

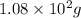
 for water =
for water = 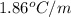
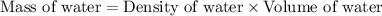
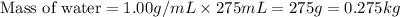
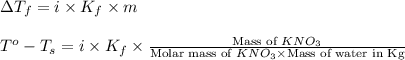
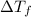 = change in freezing point
= change in freezing point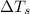 = freezing point of solution =
= freezing point of solution = 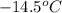
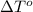 = freezing point of water =
= freezing point of water = 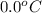
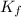 = freezing point constant for water =
= freezing point constant for water =