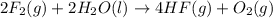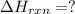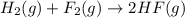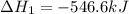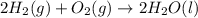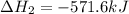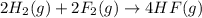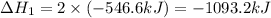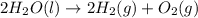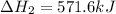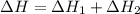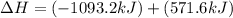Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 21:30
What is the correct name for the compound cocl3? a) cobalt(i) chloride b) cobalt(i) chlorate c) cobalt(ii) chlorate d) cobalt(iii) chloride
Answers: 1

You know the right answer?
Given that H2 (g) + F2 (g) ⟶ 2HF (g) ΔH ∘ rxn = − 546.6 kJ 2H2 (g) + O2 (g) ⟶ 2H2O (l) ΔH∘rxn = − 57...
Questions

Mathematics, 10.10.2020 16:01

History, 10.10.2020 16:01


Biology, 10.10.2020 16:01



Biology, 10.10.2020 16:01

Mathematics, 10.10.2020 16:01

Mathematics, 10.10.2020 16:01


Social Studies, 10.10.2020 16:01

Mathematics, 10.10.2020 16:01

Mathematics, 10.10.2020 16:01

English, 10.10.2020 16:01

Biology, 10.10.2020 16:01

Biology, 10.10.2020 16:01


English, 10.10.2020 16:01


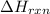 for the reaction is, -521.6 kJ
for the reaction is, -521.6 kJ