
Chemistry, 19.02.2020 03:57 dmsmith192
In a balanced chemical equation, is it true that if both reactants have an equal mass that there will be no limiting reactant? True or False and explain why

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3


Chemistry, 23.06.2019 02:00
What can be done to make a solid solute dissolve faster in a liquid solvent?
Answers: 1
You know the right answer?
In a balanced chemical equation, is it true that if both reactants have an equal mass that there wil...
Questions







Social Studies, 03.08.2019 19:00


Social Studies, 03.08.2019 19:00


Chemistry, 03.08.2019 19:00


Mathematics, 03.08.2019 19:00







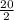 moles of O₂ = 10 mole of O₂
moles of O₂ = 10 mole of O₂

