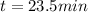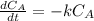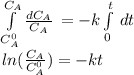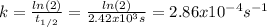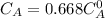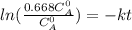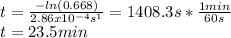Answers: 1
Another question on Chemistry

Chemistry, 20.06.2019 18:04
Can anyone (a-level) a student was analysing a carbonate compound, mco3, containing an unknown group 2 metal, m. the student carried out thermal decomposition on 0.730g of the carbonate and measured the volume of gas produced. mco3 (s) mo (s) + co2 (g) the student collected and measured 120cm3 of carbon dioxide. 1 mol of carbon dioxide occupies 24 000 cm3 under these conditions. calculate the molar mass of the group 2 carbonate and hence deduce the identity of the group 2 metal, m.
Answers: 3

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 21.06.2019 20:30
There is an area in idaho named craters of the moon where most of the ground is covered with basalt, adark gray, igneous rock with no visibl crystals. what can you infer about the geographical history of the area?
Answers: 1

Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1
You know the right answer?
The decomposition reaction of A to B is a first-order reaction with a half-life of 2.42×103 seconds:...
Questions





Health, 16.10.2019 03:50


Computers and Technology, 16.10.2019 03:50

Biology, 16.10.2019 03:50


Physics, 16.10.2019 03:50

Geography, 16.10.2019 03:50


Health, 16.10.2019 03:50

Mathematics, 16.10.2019 03:50

Mathematics, 16.10.2019 03:50

Biology, 16.10.2019 03:50




Advanced Placement (AP), 16.10.2019 03:50