
Chemistry, 21.02.2020 01:05 lucifer6669
Determine the moles of H+ reacting with the metal based on the following experimental data. 10.00 mL of 1.00 M HCl solution is added to a sample of Mg metal. The reaction goes to completion and all of the Mg metal is gone. 27.08 mL of 0.30 M NaOH solution is required to reach the end point when titrating the remaining HCl.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3


Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1
You know the right answer?
Determine the moles of H+ reacting with the metal based on the following experimental data. 10.00 mL...
Questions


History, 11.02.2021 01:00

Biology, 11.02.2021 01:00



Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

Social Studies, 11.02.2021 01:00



Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00

History, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00


Mathematics, 11.02.2021 01:00

Computers and Technology, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00


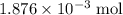 .
.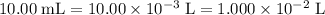 .
.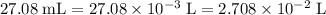 .
.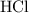 initially present (in the
initially present (in the 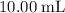 solution at
solution at 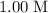 .)
.)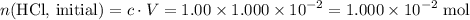 .
. from the titration:
from the titration: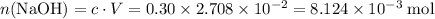 .
.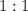 ratio:
ratio: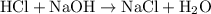 .
.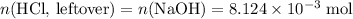 .
.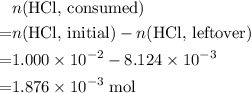 .
.

