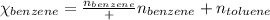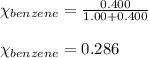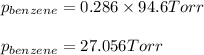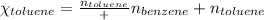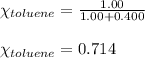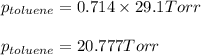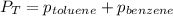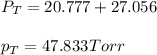Chemistry, 21.02.2020 17:21 labrandonanderson00
Benzene, C6H6, and toluene, C6H5CH3, form an ideal solution. The vapor pressure of benzene is 94.6 Torr and that of Topic 5C Exercises 372 Topic 5C Phase Equilibria in Two-Component Systems toluene is 29.1 Torr at 25 8C. What is the vapor pressure of each component at 25 8C and what is the total vapor pressure of a mix- ture of 1.00 mol benzene and 0.400 mol toluene at 25 8C?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
Alarge marble is dropped in a graduated cylinder with 35ml of water in it.the water level increases to 49ml.what is the volume of the marble
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1


Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3
You know the right answer?
Benzene, C6H6, and toluene, C6H5CH3, form an ideal solution. The vapor pressure of benzene is 94.6 T...
Questions

Mathematics, 14.12.2021 14:00

Mathematics, 14.12.2021 14:00


Mathematics, 14.12.2021 14:00

French, 14.12.2021 14:00





Mathematics, 14.12.2021 14:00


Mathematics, 14.12.2021 14:00

Computers and Technology, 14.12.2021 14:00

Computers and Technology, 14.12.2021 14:00

English, 14.12.2021 14:00


Mathematics, 14.12.2021 14:00


Mathematics, 14.12.2021 14:00

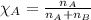
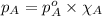 ........(1)
........(1)