
Chemistry, 22.02.2020 03:33 imogengrzemskip4rq0p
. Determine the standard free energy change, ɔ(G p for the formation of S2−(aq) given that the ɔ(G p for Ag+(aq) and Ag2S(s) are 77.1 k/mole and −39.5 kJ/mole respectively, and the solubility product for Ag2S(s) is 8 10−51.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:00
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 23.06.2019 00:30
In a ball-and-stick molecular model, what do the sticks represent?
Answers: 1

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1

Chemistry, 23.06.2019 04:20
Calculate the mass of 0.750 mol of the following substance. na3po4.
Answers: 1
You know the right answer?
. Determine the standard free energy change, ɔ(G p for the formation of S2−(aq) given that the ɔ(G p...
Questions






Mathematics, 25.12.2019 04:31














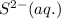 is 92.094 kJ/mol
is 92.094 kJ/mol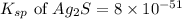
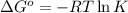
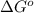 = standard Gibbs free energy = ?
= standard Gibbs free energy = ?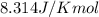
![25^oC=[273+25]K=298K](/tpl/images/0520/0871/0e82f.png)
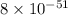
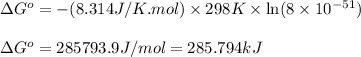
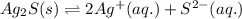
![\Delta G^o_{rxn}=\sum [n\times \Delta G^o_f_{(product)}]-\sum [n\times \Delta G^o_f_{(reactant)}]](/tpl/images/0520/0871/f2395.png)
![\Delta G^o_{rxn}=[(2\times \Delta G^o_f_{(Ag^+(aq.))})+(1\times \Delta G^o_f_{(S^{2-}(aq.))})]-[(1\times \Delta G^o_f_{(Ag_2S(s))})]](/tpl/images/0520/0871/d2b0a.png)
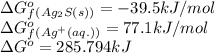
![285.794=[(2\times 77.1)+(1\times \Delta G^o_f_{(S^{2-}(aq.))})]-[(1\times (-39.5))]\\\\\Delta G^o_f_{(S^{2-}(aq.))=92.094J/mol](/tpl/images/0520/0871/afd79.png)


