(b) Magnesium reacts very slowly with cold water but it reacts faster with steam, H,0,
and for...

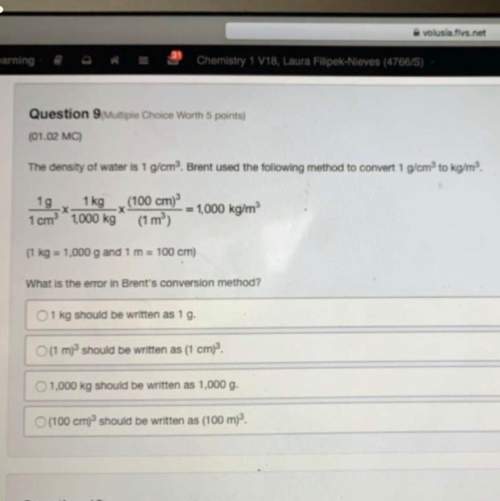
Chemistry, 23.02.2020 16:03 juicecarton
(b) Magnesium reacts very slowly with cold water but it reacts faster with steam, H,0,
and forms magnesium oxide, MgO, and hydrogen.
Write the balanced equation for the reaction between magnesium and steam.
(2)
(C) The electronic configurations of magnesium and calcium are
magnesium 2.8.2
calcium 2.8.8.2
When magnesium and calcium react with water they form positive ions.
Suggest an explanation, in terms of their electronic configurations, why calcium is
more reactive than magnesium.
(2)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:10
Identify one disadvantage to each of the following models of electron configuration: dot structures arrow and line diagrams written electron configurations type in your answer below.
Answers: 1


Chemistry, 22.06.2019 04:40
In which environment would primary succession occur? a forest with a few remaining trees after a recent wildfire an area of exposed rock after a glacier melts away beach that is exposed to the air at low tide an abandoned baseball field in a small town
Answers: 1

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2
You know the right answer?
Questions

Mathematics, 23.08.2021 06:10

Mathematics, 23.08.2021 06:10





Mathematics, 23.08.2021 06:10


Mathematics, 23.08.2021 06:10


Law, 23.08.2021 06:10


Chemistry, 23.08.2021 06:10

Mathematics, 23.08.2021 06:10

Mathematics, 23.08.2021 06:10

Mathematics, 23.08.2021 06:10

Mathematics, 23.08.2021 06:10


Social Studies, 23.08.2021 06:10