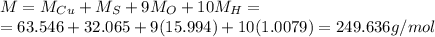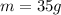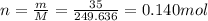Chemistry, 24.02.2020 18:06 edeliz5033
MOLES AND MASS
Name
Determine the number of moles in each of the quantities below.
1.25 g of Naci
2. 125 g of H,504
3. 100. g of KMnO,
4.'74 g of KCI
5. 35 g of CuSO,•54,0

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:00
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 04:20
Which of the following is true for the actual yield of a reaction? it is always calculated as a ratio. it is the yield from the excess reactant. it is the yield from the limiting reactant. it is always less than the theoretical yield.
Answers: 1

Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1
You know the right answer?
MOLES AND MASS
Name
Determine the number of moles in each of the quantities below.
...
Name
Determine the number of moles in each of the quantities below.
...
Questions

English, 31.07.2019 09:00

English, 31.07.2019 09:00





Social Studies, 31.07.2019 09:00







History, 31.07.2019 09:00

Mathematics, 31.07.2019 09:00

Business, 31.07.2019 09:00


English, 31.07.2019 09:00

Mathematics, 31.07.2019 09:00

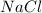 .
.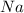 ):
): 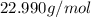
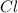 ):
): 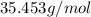
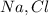 in the compound is 1:1, the molar mass of the
in the compound is 1:1, the molar mass of the 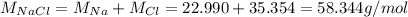
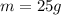
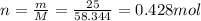
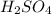
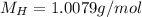 (hydrogen)
(hydrogen)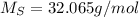 (solphur)
(solphur)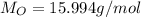 (oxygen)
(oxygen)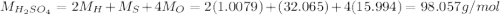
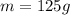
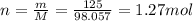
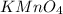
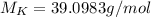 (potassium)
(potassium)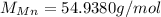 (manganese)
(manganese)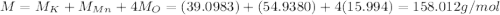
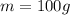
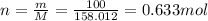
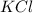
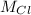 =
= 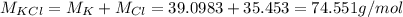
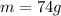
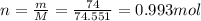
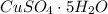
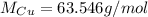 (copper)
(copper)