
Chemistry, 25.02.2020 02:52 coolcat3190
The solubility of Cr(NO3)3⋅9H2O in water is 208 g per 100 g of water at 15 ∘C. A solution of Cr(NO3)3⋅9H2O in water at 35 ∘C is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to 15 ∘C, no precipitate forms.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3


Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Chemistry, 22.06.2019 23:30
How many grams of ammonia would be produced by the decomposition of 16.93 mlof hydrazine? (the density of hydrazine is 1.021g/ml)
Answers: 3
You know the right answer?
The solubility of Cr(NO3)3⋅9H2O in water is 208 g per 100 g of water at 15 ∘C. A solution of Cr(NO3)...
Questions

Mathematics, 24.06.2019 07:20



Physics, 24.06.2019 07:20



Mathematics, 24.06.2019 07:20





Mathematics, 24.06.2019 07:20








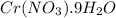 in water is 208 g per 100 g of water at
in water is 208 g per 100 g of water at 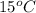 . A solution of
. A solution of 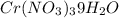 in water at
in water at 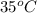 is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to
is formed by dissolving 322 g in 100 g water. When this solution is slowly cooled to 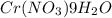 in 100 g of water = 208 g (at
in 100 g of water = 208 g (at 

