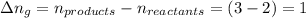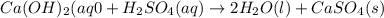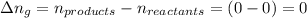Chemistry, 25.02.2020 03:04 DaFuzzyDude
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which of the following reactions will ΔH be approximately equal to ΔE? Select all that apply. Group of answer choices 2 NO2(g) -> N2(g) + 2 O2(g)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 11:00
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3
You know the right answer?
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which...
Questions





Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

English, 12.02.2021 18:30


Mathematics, 12.02.2021 18:30




Biology, 12.02.2021 18:30

History, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30

Mathematics, 12.02.2021 18:30


Mathematics, 12.02.2021 18:30

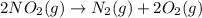
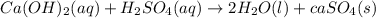
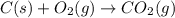
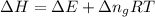 as
as 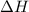 = enthalpy change
= enthalpy change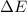 = internal energy change
= internal energy change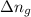 = change in number of moles of gas particles =
= change in number of moles of gas particles =