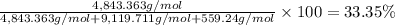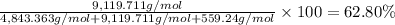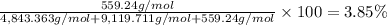Chemistry, 25.02.2020 03:52 TravKeepIt100
Convert the composition of the following alloy from atom percent to weight percent (a) 44.9 at% of silver, (b) 46.3 at% of gold, and (c) 8.8 at% of copper. The atomic weights for silver, gold, and copper are 107.87, 196.97, and 63.55 g/mol, respectively.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:50
Elements in group 2 are all called alkaline earth metals. what is most similar about the alkaline earth metals?
Answers: 1

Chemistry, 23.06.2019 01:30
Adirect relationship can be represented by: a curve a pie chart
Answers: 2

Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 1

Chemistry, 23.06.2019 14:40
Uuestons niuthe no. of millimoles of hcl required to neutralize 10 ml of 0.2 m na2co3 is(a) 2.0 m mole(b) 4.0 m mole(c) 0.2 m mole(d) 0.4 m mole
Answers: 1
You know the right answer?
Convert the composition of the following alloy from atom percent to weight percent (a) 44.9 at% of s...
Questions

English, 16.12.2020 02:30

Mathematics, 16.12.2020 02:30

Biology, 16.12.2020 02:30




History, 16.12.2020 02:30

History, 16.12.2020 02:30




Biology, 16.12.2020 02:30






Mathematics, 16.12.2020 02:30


Spanish, 16.12.2020 02:30

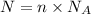
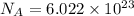 = Avogadro number
= Avogadro number