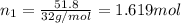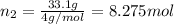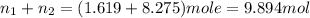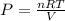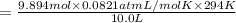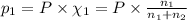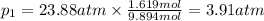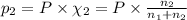Chemistry, 25.02.2020 03:50 angelteddy033
Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21 ∘C∘C. Express the pressures in atmospheres to three significant digits separated by commas.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 21:50
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2

Chemistry, 23.06.2019 05:40
Which order shows the levels of organization from largest to smallest? organism, organ system, cell, organ, tissue organism, tissue, organ system, organ, cell organism, organ, organ system, cell, tissue organism, organ system, organ, tissue, cell
Answers: 2

Chemistry, 23.06.2019 07:30
Which statement explains which thermometer is more appropriate to measure the temperature of a liquid at 43.6 degrees celsius a) thermometer a, because it measures temperature more accurately than thermometer b b) thermometer b, because it measures temperature more accurately than thermometer a c) thermometer a, because it measures temperature more precisely than thermometer b d) thermometer b, because it measures temperature more precisely than thermometer a
Answers: 2
You know the right answer?
Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 2...
Questions

Business, 02.03.2021 14:00





Physics, 02.03.2021 14:00


English, 02.03.2021 14:00

World Languages, 02.03.2021 14:00


Business, 02.03.2021 14:00

Mathematics, 02.03.2021 14:00


Mathematics, 02.03.2021 14:00


English, 02.03.2021 14:00

Mathematics, 02.03.2021 14:00

History, 02.03.2021 14:00


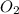 and 33.1 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21°C.Express the pressures in atmospheres to three significant digits separated by commas.
and 33.1 g of He. Calculate the partial pressure of each gas and the total pressure if the temperature of the gas is 21°C.Express the pressures in atmospheres to three significant digits separated by commas.