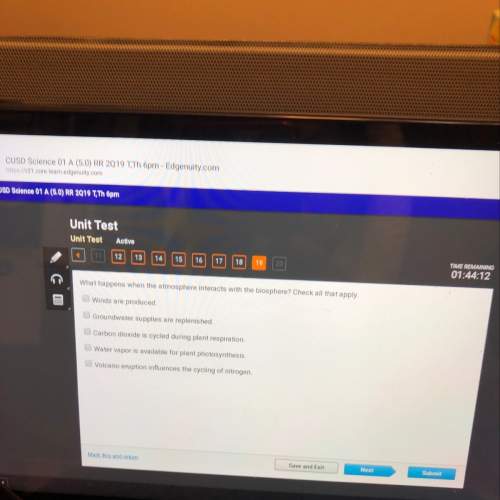
Chemistry, 25.02.2020 04:31 gianababnnna
Increasing the concentration of a reactant shifts the position of a chemical equilibrium towards formation of more products. what effect does adding a reactant have on the rates of the forward and reverse reactions?
a)There is no effect
b)Both the forward and reverse speed up by same amount
c)The foward reaction speeds up immeditaly
d)Forward reaction slows down
e)the forward reaction speeds up
f)more info needs to be given.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 05:30
What is the mass of each element in a 324.8 sample of co2
Answers: 1

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1
You know the right answer?
Increasing the concentration of a reactant shifts the position of a chemical equilibrium towards for...
Questions

Mathematics, 22.07.2019 01:40

Social Studies, 22.07.2019 01:40


Biology, 22.07.2019 01:40

Chemistry, 22.07.2019 01:40










History, 22.07.2019 01:40





English, 22.07.2019 01:40