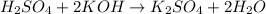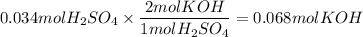The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation below. Suppose
0.06 L of KOH with unknown concentration is placed in a flask with bromthymol blue indicator. A solution of 0.20 M
H2504 is dripped into the KOH solution. After exactly 0.017 L of H2SO4 is added, the Indicator changes from blue to
yellow. What is the concentration of the KOH? You must show all of your work to earn credit. (4 points)
H2SO4 + 2KOH → K2SO4 + 2H20

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3

Chemistry, 23.06.2019 10:00
The temperature of a lead fishing weight rises from 26 °c to 38 °c as it absorbs 11.3 j of heat. what is the mass of the fishing weight in grams?
Answers: 2

Chemistry, 23.06.2019 10:00
The temperature of a lead fishing weight rises from 26 °c to 38 °c as it absorbs 11.3 j of heat. what is the mass of the fishing weight in grams?
Answers: 1
You know the right answer?
The reaction of sulfuric acid (H2SO4) with potassium hydroxide (KOH) is described by the equation be...
Questions

Mathematics, 25.06.2019 20:00

English, 25.06.2019 20:00




Mathematics, 25.06.2019 20:00

History, 25.06.2019 20:00

Mathematics, 25.06.2019 20:00

Spanish, 25.06.2019 20:00

Chemistry, 25.06.2019 20:00

Chemistry, 25.06.2019 20:00



Mathematics, 25.06.2019 20:00