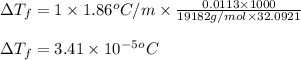A biochemical engineer isolates a bacterial gene fragment and dissolves an 11.3 mg sample of the material in enough water to make 32.2 mL of solution. The osmotic pressure of the solution is 0.340 torr at 25°C.
(a) What is the molar mass of the gene fragment?
(b) If the solution density is 0.997 g/mL, how large is the freezing point depression for this solution (Kf of water = 1.86 °C/m)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 12:00
Explaining why atoms bondcomplete the sentence.atoms form chemical bonds to satisfy the rule and to become .
Answers: 1
You know the right answer?
A biochemical engineer isolates a bacterial gene fragment and dissolves an 11.3 mg sample of the mat...
Questions


History, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Social Studies, 09.10.2019 04:30

Biology, 09.10.2019 04:30


Spanish, 09.10.2019 04:30

Health, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Health, 09.10.2019 04:30

Social Studies, 09.10.2019 04:30



Social Studies, 09.10.2019 04:30


Biology, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

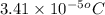
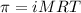
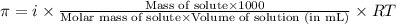
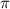 = osmotic pressure of the solution = 0.340 torr
= osmotic pressure of the solution = 0.340 torr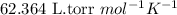
![25^oC=[273+25]=298K](/tpl/images/0523/6100/6a9f9.png)

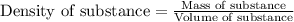
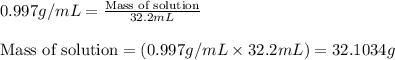
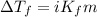
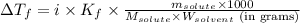
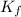 = molal freezing point elevation constant = 1.86°C/m
= molal freezing point elevation constant = 1.86°C/m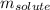 = Given mass of solute (gene fragment) = 0.0113 g
= Given mass of solute (gene fragment) = 0.0113 g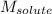 = Molar mass of solute (gene fragment) = 19182 g/mol
= Molar mass of solute (gene fragment) = 19182 g/mol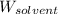 = Mass of solvent (water) = 32.0921 g
= Mass of solvent (water) = 32.0921 g