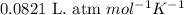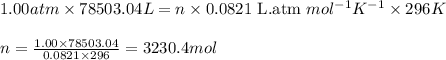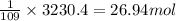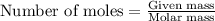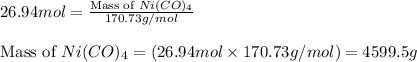Nickel carbonyl, Ni(CO)4 is one of the most toxic substances known. The present maximum allowable concentration in laboratory air during an 8-hr workday is 1 ppb (parts per billion) by volume, which means that there is one mole of Ni(CO)4 for every 109 moles of gas. Assume 23?C and 1.00 atm pressure. What mass of Ni(CO)4 is allowable in a laboratory that is 14ft X 22ft X 9ft ?

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1
You know the right answer?
Nickel carbonyl, Ni(CO)4 is one of the most toxic substances known. The present maximum allowable co...
Questions

Mathematics, 09.12.2021 01:00





History, 09.12.2021 01:00



History, 09.12.2021 01:00

Biology, 09.12.2021 01:00

History, 09.12.2021 01:00

History, 09.12.2021 01:00

Health, 09.12.2021 01:00

Social Studies, 09.12.2021 01:00

Computers and Technology, 09.12.2021 01:00


History, 09.12.2021 01:00


Business, 09.12.2021 01:00

Mathematics, 09.12.2021 01:00

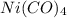 allowable in the laboratory is 4599.5 grams
allowable in the laboratory is 4599.5 grams
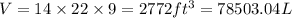 (Conversion factor:
(Conversion factor: 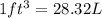
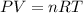
![23^oC=[23+273]K=296K](/tpl/images/0523/9842/185cf.png)