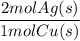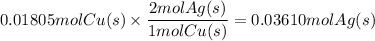Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1


Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
What mass of metallic silver can form from 1.147 g of copper metal according to equation b?...
Questions

Chemistry, 25.07.2019 03:00

English, 25.07.2019 03:00

Mathematics, 25.07.2019 03:00

Mathematics, 25.07.2019 03:00

History, 25.07.2019 03:00



Mathematics, 25.07.2019 03:00

Mathematics, 25.07.2019 03:00


English, 25.07.2019 03:00


Chemistry, 25.07.2019 03:00


English, 25.07.2019 03:00


History, 25.07.2019 03:00

Mathematics, 25.07.2019 03:00