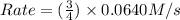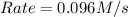Chemistry, 26.02.2020 03:09 abdullahs4639
The reaction A + B ⟶ C + D rate = k [ A ] [ B ] 2 A+B⟶C+Drate=k[A][B]2 has an initial rate of 0.0640 M / s. 0.0640 M/s. What will the initial rate be if [ A ] [A] is halved and [ B ] [B] is tripled?What will the initial rate be if [A] is tripled and [B] is halved? answer M/s

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2


Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3
You know the right answer?
The reaction A + B ⟶ C + D rate = k [ A ] [ B ] 2 A+B⟶C+Drate=k[A][B]2 has an initial rate of 0.0640...
Questions

Biology, 21.02.2021 17:10

Computers and Technology, 21.02.2021 17:10


History, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10


Mathematics, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10

Chemistry, 21.02.2021 17:10

Chemistry, 21.02.2021 17:10

Social Studies, 21.02.2021 17:10

Mathematics, 21.02.2021 17:10


Mathematics, 21.02.2021 17:10

Physics, 21.02.2021 17:10

Physics, 21.02.2021 17:10

![Rate=k[A][B]^2](/tpl/images/0524/3846/d8f90.png)
![Rate=k\times (\frac{[A]}{2})\times (3\times [B])^2](/tpl/images/0524/3846/31c7e.png)
![Rate=k\times (\frac{[A]}{2})\times 9\times [B]^2](/tpl/images/0524/3846/e768d.png)
![Rate=k\times (\frac{9}{2})\times [A]\times [B]^2](/tpl/images/0524/3846/4e890.png)
![k[A][B]^2](/tpl/images/0524/3846/8ef61.png) = 0.0640 M/s
= 0.0640 M/s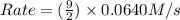
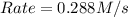
![Rate=k\times (\frac{[B]}{2})^2\times (3\times [A])](/tpl/images/0524/3846/fb264.png)
![Rate=k\times (\frac{[B]^2}{4})\times 3\times [A]](/tpl/images/0524/3846/c4659.png)
![Rate=k\times (\frac{3}{4})\times [A]\times [B]^2](/tpl/images/0524/3846/8107a.png)