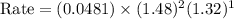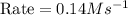For the reaction, A(g) + B(g) => AB(g), the rate is 0.385 mol/L. s when the initial concentrations of both A and B are 2.00 mol/L. If the reaction is second order in A and first order in B, what is the rate when the initial concentration of A = 1.48 mol/L and that of B = 1.32 mol/L. Give your answer to 2 decimal places

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1



Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3
You know the right answer?
For the reaction, A(g) + B(g) => AB(g), the rate is 0.385 mol/L. s when the initial concentration...
Questions

History, 03.02.2020 15:43

Mathematics, 03.02.2020 15:43

Biology, 03.02.2020 15:43

English, 03.02.2020 15:43



Mathematics, 03.02.2020 15:43

Mathematics, 03.02.2020 15:43

History, 03.02.2020 15:43

Physics, 03.02.2020 15:43

History, 03.02.2020 15:43


Mathematics, 03.02.2020 15:43

Geography, 03.02.2020 15:43






Mathematics, 03.02.2020 15:43

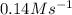
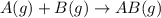
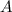 and
and 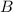 are the reactants.
are the reactants.![\text{Rate}=k[A]^2[B]^1](/tpl/images/0524/7200/858f4.png)
![\text{Rate}=k[A]^2[B]](/tpl/images/0524/7200/15407.png)
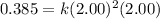
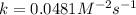
![\text{Rate}=k[A]^2[B]^0[C]^1](/tpl/images/0524/7200/54afd.png)