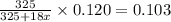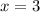Chemistry, 26.02.2020 18:29 unknown337
4. A 120.0 mg sample of a lead acetate hydrate, Pb(C2H3O2)2 × xH2O was heated to drive off the waters of hydration. The cooled residue had a mass of 103.0 mg. Calculate the value of x in the chemical formula. Show your work with Equation Editor.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2
You know the right answer?
4. A 120.0 mg sample of a lead acetate hydrate, Pb(C2H3O2)2 × xH2O was heated to drive off the wate...
Questions

Mathematics, 31.03.2021 04:00

Mathematics, 31.03.2021 04:00

Social Studies, 31.03.2021 04:00

Mathematics, 31.03.2021 04:00




English, 31.03.2021 04:00







Mathematics, 31.03.2021 04:00


History, 31.03.2021 04:00

Mathematics, 31.03.2021 04:00

History, 31.03.2021 04:00

Mathematics, 31.03.2021 04:00

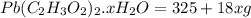
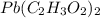 = 325 g
= 325 g
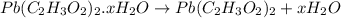
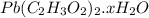 on heating gives = 325 g of
on heating gives = 325 g of 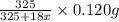 of
of