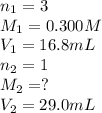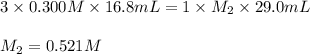Ou are titrating a solution of sodium hydroxide of unknown concentration with a solution of phosphoric acid that has a concentration of 0.300 M. Starting with 29.0 mL of the sodium hydroxide solution, youuse 16.8 mL of the acid to titrate the base to the endpoint. Calculate the concentration of the sodium hydroxide solution.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
Ou are titrating a solution of sodium hydroxide of unknown concentration with a solution of phosphor...
Questions

Business, 21.09.2019 00:30


English, 21.09.2019 00:30



Spanish, 21.09.2019 00:30

Mathematics, 21.09.2019 00:30



Biology, 21.09.2019 00:30

Mathematics, 21.09.2019 00:30



Mathematics, 21.09.2019 00:30

Business, 21.09.2019 00:30


Mathematics, 21.09.2019 00:30


Physics, 21.09.2019 00:30

Mathematics, 21.09.2019 00:30

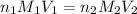
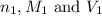 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 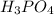
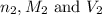 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.