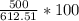Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 22:30
Astudent pours 10.0 g of salt into a container of water and observes the amount of time it takes for the salt to dissolve. she then repeats the process using the same amounts of salt and water but this time she slowly stirs the mixture while it is dissolving. the student performs the experiment one more time but this time she stirs the mixture rapidly. the dependent variable in this experiment is: time for salt to dissolve speed of stirring amount of water mass of salt
Answers: 1

Chemistry, 23.06.2019 04:20
The equation below shows the reaction of zinc with hydrochloric acid (hcl). zn (s) + 2 hcl (aq) —> zncl2 (aq) + h2 (g) what will happen if the concentration of hcl is decreased? a. more zncl2 will be produced. b. the reaction rate will slow down. c. the hydrochloric acid will become more acidic. d. the reaction will produce water instead of hydrogen gas.
Answers: 1

Chemistry, 23.06.2019 09:00
The concentration of ionic substances is important for the heart to beat. your heart responds to electrical impulses that travel through heart cells that are made up mostly of water. which properties of ionic compounds are important to support this function? solubility in water conductivity crystalline melting point
Answers: 3

Chemistry, 23.06.2019 11:20
Ajar is tightly sealed at 22°c and 772 torr what is the pressure inside a jar after its been heated to 178°c
Answers: 1
You know the right answer?
For the reaction represented by the equation 1SO3 1H2O -> 1H2SO4, calculate the percentage yield...
Questions


Mathematics, 27.09.2020 09:01

Mathematics, 27.09.2020 09:01

English, 27.09.2020 09:01

History, 27.09.2020 09:01




Computers and Technology, 27.09.2020 09:01


Mathematics, 27.09.2020 09:01

Mathematics, 27.09.2020 09:01

Mathematics, 27.09.2020 09:01

Geography, 27.09.2020 09:01


Social Studies, 27.09.2020 09:01




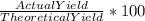 =
=