Can someone help me complete this table and find actual and theoretical yield
...

Chemistry, 27.02.2020 02:23 ashlynnplace
Can someone help me complete this table and find actual and theoretical yield
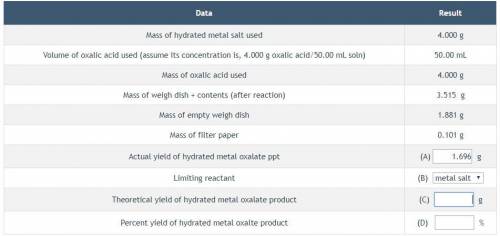

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:40
Effect of rotenone and antimycin a on electron transfer rotenone, a toxic natural product from plants, strongly inhibits nadh dehydrogenase of insect and fish mitochondria. antimycin a, a toxic antibiotic, strongly inhibits the oxidation of ubiquinol. (a) explain why rotenone ingestion is lethal to some insect and fish species. (b) explain why antimycin a is a poison. (c) given that rotenone and antimycin a are equally effective in blocking their respective sites in the electron-transfer chain, which would be a more potent poison? explain.
Answers: 3

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3
You know the right answer?
Questions

History, 16.02.2021 18:30


English, 16.02.2021 18:30

English, 16.02.2021 18:30

Mathematics, 16.02.2021 18:30


History, 16.02.2021 18:30

Mathematics, 16.02.2021 18:30

Mathematics, 16.02.2021 18:30


Mathematics, 16.02.2021 18:30

Computers and Technology, 16.02.2021 18:30



Mathematics, 16.02.2021 18:30

History, 16.02.2021 18:30


Mathematics, 16.02.2021 18:30


Social Studies, 16.02.2021 18:30



