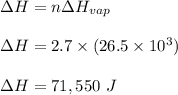Chemistry, 27.02.2020 04:35 walmartislife
Calculate the change in entropy that occurs in the system when 2.70 mole of diethyl ether (C4H6O) condenses from a gas to a liquid at its normal boiling point (34.6∘C). ΔHvap = 26.5 kJ/mol

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1


Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3
You know the right answer?
Calculate the change in entropy that occurs in the system when 2.70 mole of diethyl ether (C4H6O) co...
Questions

History, 28.09.2021 07:00

Mathematics, 28.09.2021 07:00

History, 28.09.2021 07:00


Mathematics, 28.09.2021 07:10



Mathematics, 28.09.2021 07:20



English, 28.09.2021 07:20

Mathematics, 28.09.2021 07:30

World Languages, 28.09.2021 07:30



Mathematics, 28.09.2021 07:30

Mathematics, 28.09.2021 07:30