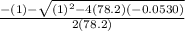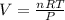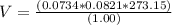Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g) is placed in the cylinder represented below. The barometric pressure on the piston (which is assumed to be massless and frictionless) is constant at 1.00 atm. The original volume (before the 0.386 mol C(g) begins to decompose) is 7.29 L. The temperature is fixed and can be determined from the idea gas law. What is the volume in the cylinder at equilibrium?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1

Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1
You know the right answer?
Consider the reaction below for which K = 78.2 atm-1. A(g) + B(g) ↔ C(g) Assume that 0.386 mol C(g)...
Questions

English, 18.09.2019 11:00


Spanish, 18.09.2019 11:00

Mathematics, 18.09.2019 11:00


Biology, 18.09.2019 11:00



Advanced Placement (AP), 18.09.2019 11:00


Social Studies, 18.09.2019 11:00


Chemistry, 18.09.2019 11:00

Social Studies, 18.09.2019 11:00

Computers and Technology, 18.09.2019 11:00


History, 18.09.2019 11:00


Biology, 18.09.2019 11:00

History, 18.09.2019 11:00

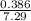
![K = \frac{[C]}{[A][B]}](/tpl/images/0526/7975/63063.png)
![78.2=\frac{[0.0530-x]}{[x][x]}](/tpl/images/0526/7975/8334d.png)
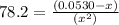
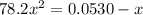
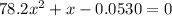
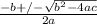
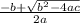 or
or 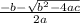
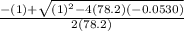 or
or