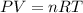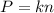Two flasks of equal volume and at the same temperature contain different gases. One flask contains 10.0 g of Ne, and the other flask contains 10.0 g of He. Is each of the following statements true or false? Explain.1) True. Because the gases have the same mass, they exert the same pressures.2) True. Both the volumes and the temperatures of the gases are the same, which means that their pressures must be equal.3) False. There are different numbers of moles in the flasks, meaning that the pressures are different.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 16:00
He table below gives the atomic mass and relative abundance values for the three isotopes of element m. relative abundance (%) atomic mass (amu) 78.99 23.9850 10.00 24.9858 11.01 25.9826 what is the average atomic mass (in amu) of element m? 2.86 5.36 24.30 24.98
Answers: 2
You know the right answer?
Two flasks of equal volume and at the same temperature contain different gases. One flask contains 1...
Questions

Social Studies, 13.11.2020 22:10


Mathematics, 13.11.2020 22:10

Biology, 13.11.2020 22:10



Mathematics, 13.11.2020 22:10

Mathematics, 13.11.2020 22:10


Mathematics, 13.11.2020 22:10




Mathematics, 13.11.2020 22:10

English, 13.11.2020 22:10



Mathematics, 13.11.2020 22:10

Mathematics, 13.11.2020 22:10

Chemistry, 13.11.2020 22:10