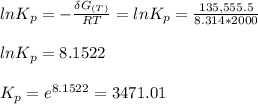Chemistry, 28.02.2020 02:03 Lizzy527663
Calculate the equilibrium composition for the reaction H2−− 1 2O2 −−)−−* H2O when the ratio of the number of moles of elemental hydrogen to elemental oxygen is unity. Perform this calculation at T = 2000 K and P = 1 atm.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 08:30
Agroup of students is studying convection current. they fill two identical balloons with the same amount of helium. one balloon is placed in a freezer and the other is in an area with warm air. after 10 minutes, the balloon are released from a height of 1 meter. which of the following to the students most likely observe? a) the warm balloon expands and rises. the cold balloon shrinks and sinks b) the balloon both rise. the cold balloon is larger than the warm balloon c) the cold balloon expands and rises. the warm balloon shrinks and sinks d) the balloon rise at the same rate. both balloons are the same size
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
Calculate the equilibrium composition for the reaction H2−− 1 2O2 −−)−−* H2O when the ratio of the n...
Questions

History, 06.05.2020 21:21


Mathematics, 06.05.2020 21:21


Mathematics, 06.05.2020 21:21



Mathematics, 06.05.2020 21:21



Mathematics, 06.05.2020 21:21





History, 06.05.2020 21:21

History, 06.05.2020 21:21



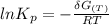 ---------(i)
---------(i)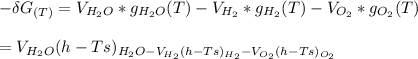
![= V_{H_20[h_f+h_{2000}-hs)-Ts]H_2O - V_{H_2}[h_f+h_{2000}-hs)-Ts]H_2- V_{O_2}[h_f+h_{2000}-hs)-Ts]O_2\\](/tpl/images/0527/5740/e8071.png)