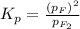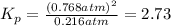At 500 degree C, F_2 gas is stable and does not dissociate, but at 840 degree C, some dissociation occurs: F_2 (g) 2 F(g). A flask filled with 0.600 atm of F_2 at 500 degree C was heated to 840 degree C, and the pressure at equilibrium was measured to be 0.984 atm. What is the equilibrium constant K_p for the dissociation of F_2 gas at 840 degree C?

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1
You know the right answer?
At 500 degree C, F_2 gas is stable and does not dissociate, but at 840 degree C, some dissociation o...
Questions


History, 07.05.2021 01:00


Computers and Technology, 07.05.2021 01:00

Spanish, 07.05.2021 01:00

Mathematics, 07.05.2021 01:00





Computers and Technology, 07.05.2021 01:00

Chemistry, 07.05.2021 01:00

Mathematics, 07.05.2021 01:00

History, 07.05.2021 01:00


Mathematics, 07.05.2021 01:00




Mathematics, 07.05.2021 01:00

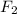 gas at 840 degree Celsius.
gas at 840 degree Celsius.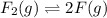
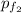 = (0.600 atm - 0.384 atm)=0.216 atm
= (0.600 atm - 0.384 atm)=0.216 atm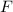 gas,
gas, 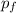 = 2(0.384 atm)=0.768 atm
= 2(0.384 atm)=0.768 atm