
Chemistry, 28.02.2020 19:11 sadmomsclub
Suppose the first equation is reversed and multiplied by 1/6, the second and third equations are divided by 2, and the three adjusted equations are added. What is the net reaction?3 A + 6 B → 3 D, ΔH = -402 kJ/molE + 2 F → A, ΔH = -108.3 kJ/molC → E + 3 D, ΔH = +65.5 kJ/mol

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 2

Chemistry, 23.06.2019 03:00
Analyze the reaction to determine whether the reaction is exothermic or endothermic. explain your reasoning.
Answers: 1

Chemistry, 23.06.2019 05:50
Which of the following is not a characteristic of s waves?
Answers: 1

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
You know the right answer?
Suppose the first equation is reversed and multiplied by 1/6, the second and third equations are div...
Questions














Computers and Technology, 06.07.2021 22:30


Mathematics, 06.07.2021 22:30

Mathematics, 06.07.2021 22:30

Mathematics, 06.07.2021 22:30


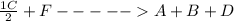 ;ΔH = + 45.6 kJ/mol
;ΔH = + 45.6 kJ/mol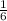 ; we have:
; we have: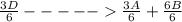 ; Δ
; Δ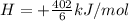
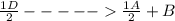 ; Δ
; Δ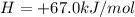 ----Equation 1
----Equation 1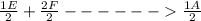 ;ΔH =
;ΔH = 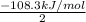
 ;ΔH = -54.15 kJ/mol --- Equation 2
;ΔH = -54.15 kJ/mol --- Equation 2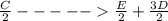 ;ΔH =
;ΔH = 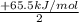
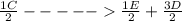 ;ΔH = + 32.75 kJ/mol --- Equation 3
;ΔH = + 32.75 kJ/mol --- Equation 3

