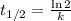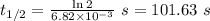Chemistry, 28.02.2020 19:45 brianadee800
The first-order rate constant for the decomposition of N2O5, 2N2O5(g)→4NO2(g)+O2(g) at 70∘C is 6.82×10−3 s−1. Starting with 8.00×10−2 mol of N2O5(g) in a volume of 2.9 L, how many moles of reactant are left after 5 minutes? What is its half-life?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
1. baking powder is a 1: 1 molar mixture of cream of tartar (khc4h4o6) and baking soda (nahco3). a recipe calls for two teaspoons (a total of 8.0 grams) of cream of tartar. how much baking soda must be added for both materials to react completely?
Answers: 2

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

You know the right answer?
The first-order rate constant for the decomposition of N2O5, 2N2O5(g)→4NO2(g)+O2(g) at 70∘C is 6.82×...
Questions

Mathematics, 22.02.2021 23:30


History, 22.02.2021 23:30


Mathematics, 22.02.2021 23:30

Mathematics, 22.02.2021 23:30

Engineering, 22.02.2021 23:30






Chemistry, 22.02.2021 23:30





Social Studies, 22.02.2021 23:30

Mathematics, 22.02.2021 23:30

![[A_t]=[A_0]e^{-kt}](/tpl/images/0528/2808/1ef89.png)
![[A_t]](/tpl/images/0528/2808/5262c.png) is the concentration at time t
is the concentration at time t ![[A_0]](/tpl/images/0528/2808/9a686.png) is the initial concentration =
is the initial concentration = 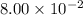 mol
mol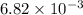 s⁻¹
s⁻¹![[A_t]=8.00\times 10^{-2}e^{-6.82\times 10^{-3}\times 300}\ mol=0.01034\ mol](/tpl/images/0528/2808/69780.png)