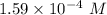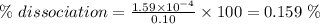An acid with the general formula RCOOH is used to make a 0.10 M solution in water. The pH of this acid solution is measured as 3.8. Calculate the extent of dissociation of this acid in the prepared solution. (The extent of dissociation is the fraction of the acid that is dissociated into the RCOO- anion and H3O cation in the aqueous solution.)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2



Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2
You know the right answer?
An acid with the general formula RCOOH is used to make a 0.10 M solution in water. The pH of this ac...
Questions

Mathematics, 04.10.2019 18:30


Mathematics, 04.10.2019 18:30






Physics, 04.10.2019 18:30

Social Studies, 04.10.2019 18:30


Mathematics, 04.10.2019 18:30








Social Studies, 04.10.2019 18:30

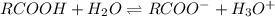
![[H^+]](/tpl/images/0528/9628/07acb.png)