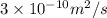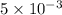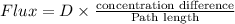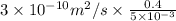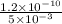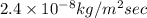A plate of iron is exposed to a carbon-rich (carburizing) atmosphere on one side and a carbon-deficient atmosphere on the other side at 1200K. If a condition of steady state is achieved, calculate the diffusion flux of carbon through the plate if the concentrations of carbon at positions of 5 and 10 mm beneath the carburizing surface are 1.2 and 0.8 kg/m3, respectively. Assume the diffusion coefficient is 3×10-10 m2/s at this temperature.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 07:10
Provide a stepwise curved arrow mechanism that fully explains the outcome of the reaction shown below. oh нао* heat он
Answers: 2

Chemistry, 22.06.2019 10:30
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3
You know the right answer?
A plate of iron is exposed to a carbon-rich (carburizing) atmosphere on one side and a carbon-defici...
Questions


Chemistry, 13.07.2020 18:01

Mathematics, 13.07.2020 18:01



Mathematics, 13.07.2020 18:01


Biology, 13.07.2020 18:01

Mathematics, 13.07.2020 18:01



Health, 13.07.2020 18:01

Mathematics, 13.07.2020 18:01





Mathematics, 13.07.2020 18:01


Mathematics, 13.07.2020 18:01